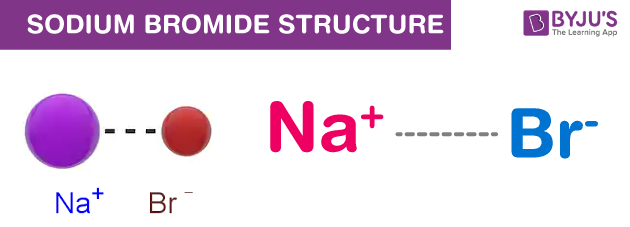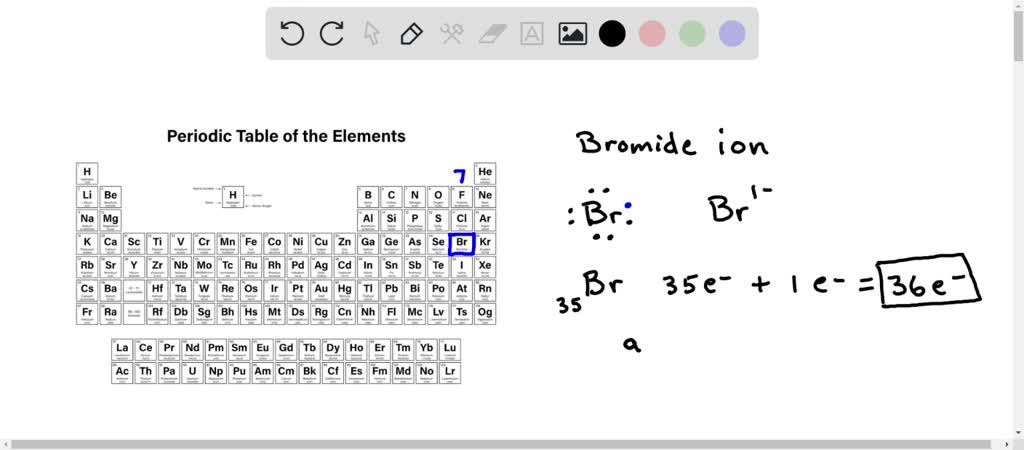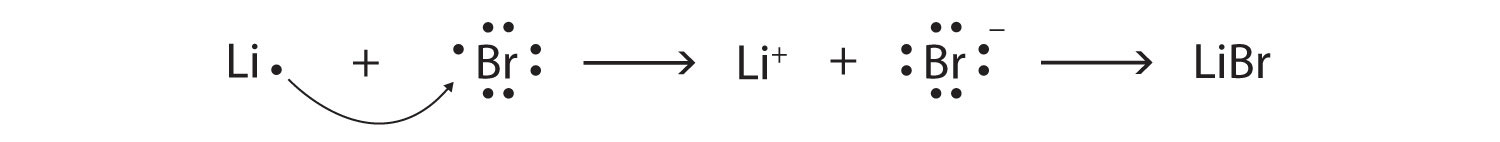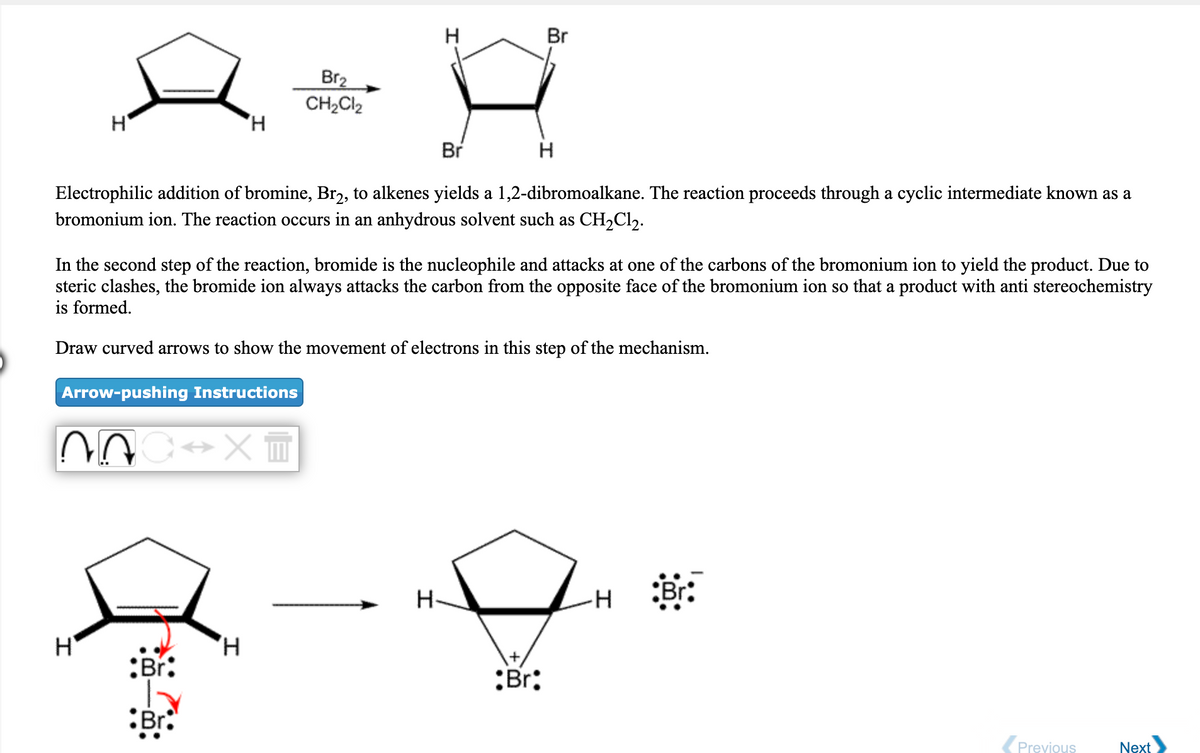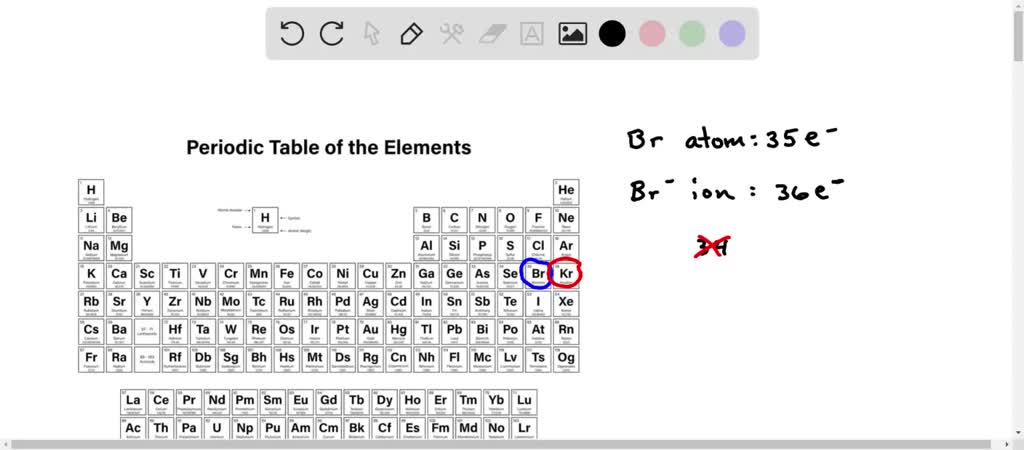
SOLVED: Bromine tends to form simple ions which have the electronic configuration of a noble gas. What is the electronic configuration of the noble gas which the bromide ion mimics? Group of

Permanganate ion reacts with bromide ion in basic medium to give magnesium dioxide and bromate ion.Write the balanced ionic equation for the reaction.
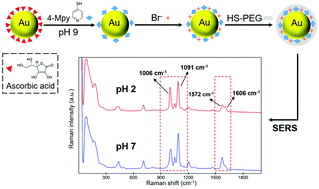
Bromide ion-functionalized nanoprobes for sensitive and reliable pH measurement by surface-enhanced Raman spectroscopy - Analyst (RSC Publishing)

SOLVED: What is the concentration of an AlBr3 solution if 150.mL of the solution contains 250.mg of Br" ion? OA) 1.67M B) 6.23 10-3 M 2.08 x 10-2 M D) 6.95 x 10 3 M
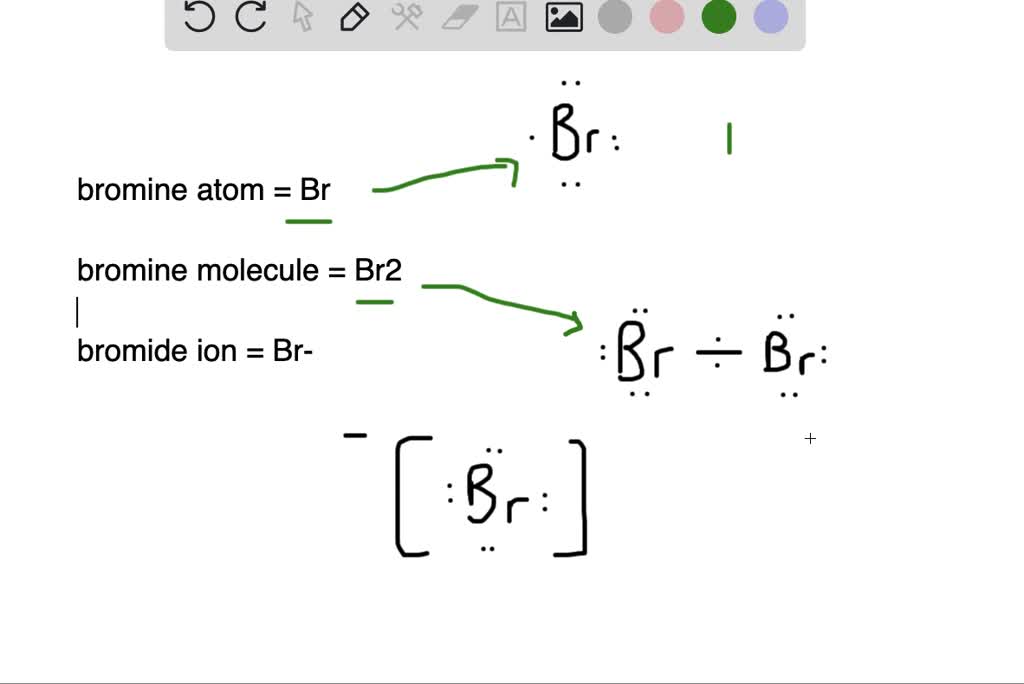
SOLVED:What is the difference between (a) a bromine atom, (b) a bromine molecule, and (c) a bromide ion? Draw the Lewis structure for each.

Question Video: Writing a Net Ionic Equation for the Reaction of Chlorine Water with Aqueous Sodium Bromide | Nagwa